www.drjwv.com
Health Issues:
Written by: Jeff Vidt, DVM
 The Answer Continued
The Answer Continued
Way back in 1983 we first started recognizing episodic fevers in Shar-Pei often associated with a swollen, painful hock joint. Later on it was observed that an early-onset kidney failure occurred in many of these dogs as well. I remember many individuals, including myself, remarking that this has to be associated with the wrinkles in some way and now we know it does.
An important article was recently published in March, 2011, entitled “A Novel Unstable Duplication Upstream of HAS2 Predisposes to a Breed-Defining Skin Phenotype and a Periodic Fever Syndrome in Chinese Shar-Pei Dogs”, Mia Olsson et al. It is the product of a consortium of researchers from several countries, working in several laboratories and our own Dr. Linda Tintle. http://www.plosgenetics.org/article/info:doi/10.1371/journal.pgen.1001332
The major points of the article are as follows:
- Chromosome 13
- Using a genome-wide analysis of Shar-Pei and other breeds a breed-specific signal occurred on chromosome 13.
- That same region had the strongest association for susceptibility to FSF.
- Two partially overlapping duplications 14.3 kb and 16.1 kb in size were located which are unique to Shar-Pei and upstream from the HAS2 gene.
- HAS2
- This gene encodes the rate-limiting enzyme synthesizing hyaluronan (HA).
- HA is up-regulated in the folded skin of the Shar-Pei. This is reflected in the higher levels of serum hyaluronan seen in the majority of the breed.
- 16.1 kb duplication
- A high copy number of this duplication results in
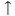 expression of HAS2 and
expression of HAS2 and 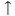 incidence of FSF and/or amyloidosis. It appears this region has a regulatory function and the mutation results in over expression of HAS2.
incidence of FSF and/or amyloidosis. It appears this region has a regulatory function and the mutation results in over expression of HAS2.
- Hyper HA syndrome
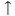 copy number variation (CNV) in this mutation predisposes these dogs to FSF.
copy number variation (CNV) in this mutation predisposes these dogs to FSF.- Identification of HA as a major risk factor for FSF (and human periodic fevers?).
- Cutaneous mucinosis (Hyper HA syndrome) and FSF are connected and have the same genetic origin – a regulatory gene mutation located close to HAS2.
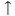 HA responsible for the skin condition (excessive folds) predisposes to sterile fever and inflammation.
HA responsible for the skin condition (excessive folds) predisposes to sterile fever and inflammation.
- Copy number
- A high copy number is not just a genetic marker for FSF but is causally related to the development of disease.
- A higher copy number enhanced the appearance of the phenotype.
- Lowest copy number in Shar-Pei is 5 and the highest is 15 (so far).
- < 4 copies – OK
- 5-10 copies - increased risk of FSF
- > 10 copies - increased risk of FSF and amyloidosis
- 2 alleles involved – total copy number of both alleles
- The mutation occurs as a single copy in other dog breeds (it is a derived mutation in Shar-Pei).
- It is being called “the meatmouth Shar-Pei duplication” because the 16.1 kb duplication appears only in meatmouth Shar-Pei – a type which has elevated levels of HA compared to both traditional Shar-Pei and other breeds.
- Alterations in balance between native high molecular weight HA vs. fragmented
low molecular weight HA (including oligomers) may result in activation of the innate immune system.
- Two pathways lead to increased formation of IL-1β, a pro-inflammatory cytokine.
- One pathway involves Toll-like receptors TLR2 and TLR4.
- The other pathway involves the NLRP3 inflammasome – a NLRP3 scaffold, ASC adaptor protein and caspase-1 enzyme. This inflammasome is present in the cytoplasm of monocytes, macrophages and mast cells.
- It is likely a blood test may be available in the near future to screen Shar-Pei for
their CNV number and the meatmouth mutation. Breeders can avoid using those individuals with high CNV numbers in their breeding programs. This test may also be useful in the general Shar-Pei population to pick out those individuals who may be at risk for FSF +/- amyloidosis. A possible blood test is being validated at this time.
- A more general conclusion from this paper is the association of degree of
wrinkling in American Shar-Pei with the incidence of FSF and amyloidosis. A general guideline would be to only use those individuals with mild to moderate wrinkling in breeding programs and breed away from Hyper HA dogs. It would appear these guidelines are not necessary in the traditional Chinese Shar-Pei.
- Lastly, the association of over-expression of HAS2 and autoinflammation most certainly has implications in the periodic fever syndromes in humans many of which are unexplained at this time.
Jeff Vidt, DVM (3/20/11)
 The Answer Continued
The Answer Continued The Answer Continued
The Answer Continued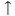 expression of HAS2 and
expression of HAS2 and 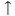 incidence of FSF and/or amyloidosis. It appears this region has a regulatory function and the mutation results in over expression of HAS2.
incidence of FSF and/or amyloidosis. It appears this region has a regulatory function and the mutation results in over expression of HAS2. 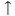 copy number variation (CNV) in this mutation predisposes these dogs to FSF.
copy number variation (CNV) in this mutation predisposes these dogs to FSF.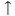 HA responsible for the skin condition (excessive folds) predisposes to sterile fever and inflammation.
HA responsible for the skin condition (excessive folds) predisposes to sterile fever and inflammation.