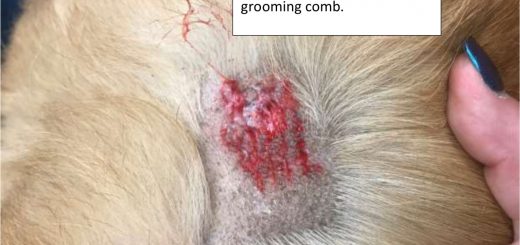
Skin Tears
I primarily associate skin tears in the American standard Chinese Shar-Pei with the prominent skin folds, “meat mouth” muzzle and thicker skin with bullae (blisters/vesicles). The skin often resembles the texture of an orange peel. This is known as Hereditary Cutaneous Mucinosis (Hyaluronosis). This condition results in high levels of hyaluronan in the dermis and epidermis of the skin. The excess hyaluronan disrupts the organization of collagen fibers in the dermis which are responsible for the strength and support of the skin much as iron rebar or steel mesh is used to strengthen concrete driveways and patios. Lastly, the excess...